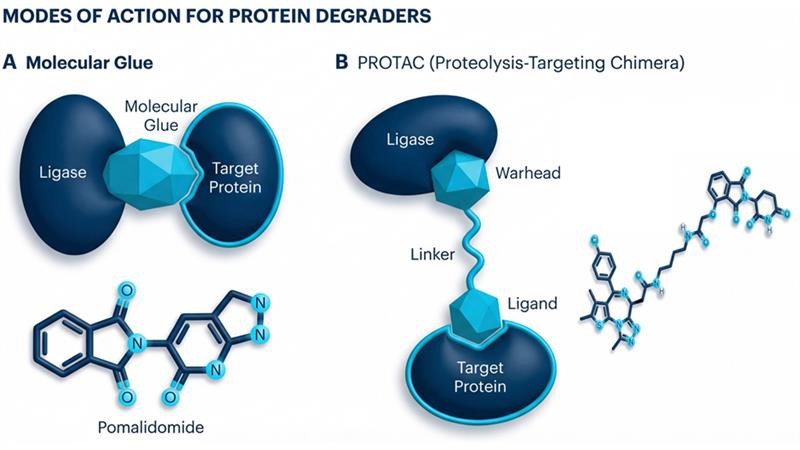
PROTACs and Molecular Glues
A single PROTAC molecule has two different binding ligands attached by a linker. One ligand binds to the target protein and the other recruits an E3 ubiquitin ligase, a cellular enzyme that marks proteins for destruction.
The PROTAC brings the E3 ligase into close contact with the target protein, forming a stable tertiary complex. The E3 ligase then attaches a chain of ubiquitin molecules to the target protein. This ubiquitin tag signals to the cells proteome to recognise and break down the tagged target protein into its basic building blocks.
After the target protein is degraded, the PROTAC molecule detaches and can then bind to another target protein, starting the cycle again, allowing for lower doses and prolonged effects.
Molecular Glues are small molecules that induce or stabilise interactions between two proteins (often a target protein and an E3 ubiquitin ligase), that otherwise would not bind. They work by binding to a primary protein, altering the surface to create a “neoepitope”, which then allows a secondary protein to bind forming a stable ternary complex.
The most common application involves hijacking the ubiquitin-proteasome system. The glue bridges an E3 ligase with a pathogenic target protein, leading to its ubiquitination and subsequent degradation by the proteosome.
Molecular glues work by directly modulating or re-enforcing existing, weak protein-protein interactions or enabling entirely new interactions. Unlike PROTACS, which use a linker, molecular glues act allosterically, with a small molecule, typically less than 500 Da, sitting directly in the interface between two proteins.
Regulated Bioanalysis of Protein Degraders
PROTACs and molecular glues are ideal in molecular weight for quantification by LC-MS/MS. The glues are the size of traditional small molecules and do not pose any additional bioanalytical challenges.
PROTACs are often described as high molecular weight small molecules with typical masses of between 700 and 1200 Da, extending up to 1500 Da in some cases. Typical extraction techniques include the use of 96 well plate protein precipitation/phospholipid removal plates for early phase studies with weak anion exchange solid phase extraction (SPE) being the best option in later phase clinical studies. Using SPE ensures better sample recoveries with lower matrix effects and enables concentration of the sample without the need for blow down and reconstitution.
Care must be taken during extraction and analysis to avoid non-specific binding. The use of low bind extraction tubes and injection plates is recommended along with the Waters Acquity Premier UPLC system.
Mass spectrometry parameters should be optimised carefully, with PROTACs often benefiting from lower ionisation energies.
Pomalidomide Case Study
The LC-MS/MS team at Synexa are currently supporting a phase 1/11a trial involving the quantification of pomalidomide, a molecular glue, in human plasma for pharmacokinetic analysis.
The method was fully validated to M10 guidelines over the calibration range 0.5 to 100 ng/ml.
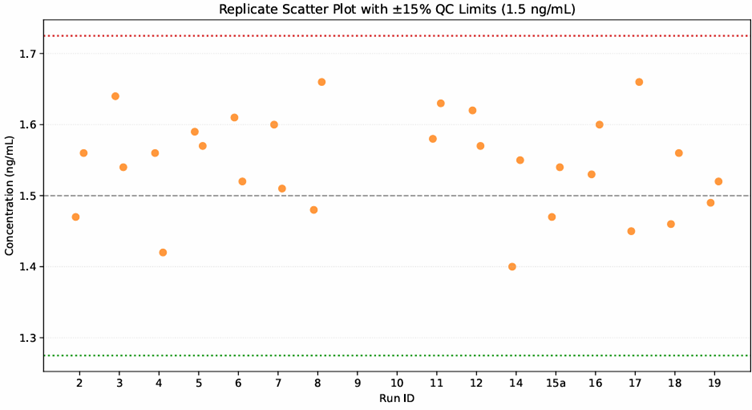
The study is on-going with 18 bioanalytical batches run to date. The assay is performing well with all quality control samples within acceptable limits and an incurred sample analysis (ISR) pass rate of 100%.
The plot below shows the low QC data from the study so far.
About Synexa Life Sciences
At Synexa, our UK site is equipped with state-of-the-art UPLC-MS/MS platforms, ideally positioned just 15 minutes from Manchester Airport to support rapid turnaround for clinical studies. Operating from a GLP/GCP-accredited facility, we provide seamless bioanalytical and biomarker support throughout the entire drug development continuum – from pre-clinical to clinical phases.
Beyond LC-MS/MS, our comprehensive analytical capabilities, including ligand binding and flow cytometry assays, enable in-depth insights into post-translational modifications and other complex biological processes.
If you would like to learn more about how we can support your bioanalysis or biomarker needs, visit www.synexagroup.com.

Written by Sally Hannam, Scientific Director
SHannam@synexagroup.com
contactus@synexagroup.com
www.synexagroup.com
FAQ
How does Synexa approach the bioanalysis of PROTACs given their higher molecular weights and structural complexity?
At Synexa, PROTAC bioanalysis is tailored to address their larger molecular weights and increased hydrophobicity, which can introduce non‑specific binding and reduced ionisation efficiency. We utilise appropriate extraction techniques, such as protein precipitation or phospholipid removal early on, moving to weak anion exchange SPE for later‑phase studies, to improve recovery and manage matrix effects. Our LC‑MS/MS workflows also incorporate careful optimisation of ionisation parameters to ensure sensitive and reproducible quantification. This enables robust data generation across both discovery and clinical phases.
How does Synexa minimise non‑specific binding during PROTAC extraction and analysis?
Synexa reduces non‑specific binding by using low‑bind tubes, plates, and consumables that help maintain analyte integrity throughout extraction and injection. PROTACs can adhere strongly to common laboratory plastics, so using specialised materials is essential to maintain consistent recovery. We also leverage high‑performance systems such as the Acquity Premier UPLC to limit surface interactions within the analytical path. These measures collectively support precise, reliable quantification.
What experience does Synexa have supporting regulated studies involving protein degraders?
Synexa is currently supporting a Phase I/IIa clinical study quantifying pomalidomide, a molecular glue, in human plasma, demonstrating our applied expertise with regulated TPD bioanalysis. The method is fully validated under ICH M10 guidelines and has delivered strong performance across 18 batches, including a 100% ISR pass rate. This real‑world experience reflects our ability to manage complex modalities under GLP/GCP conditions. Our UK LC‑MS/MS facility is equipped to deliver rapid, high‑quality support for ongoing clinical development.




