SARS CoV-2: when does the calm become the perfect storm?
BY
Prof. Patrick JD Bouic
CATEGORIES
TAGS
BY
Prof. Patrick JD Bouic
SARS-CoV-2, the causative agent of the Covid-19 pandemic, has held the globe in a total stranglehold since it’s first appearance, sweeping through nation after nation, leading to economies collapsing and numerous deaths worldwide. This is surprising since it appears that our immune system is well-equipped to deal with this virus, with the vast majority of patients remaining relatively, or even completely asymptomatic. However, there is a subset of patients that become infected and progress with severe complications, requiring hospitalization and, for some, even ICU admission.
The prognosis of hospitalized patients remains poor due to a so-called cytokine storm, the presence of which is evident according to both haematological and biochemical parameters. To date, science is unable to explain the wide spectrum of clinical responses to the viral infection. Why is there then a period of total calm as the person deals with the viral infection and yet unaware of the storm to follow? What early biomarkers of disease complications could one monitor for the eventual “perfect storm” looming on the horizon and can we intervene to deviate from the perilous storm to a more manageable rough sea?
Numerous studies have reported on the observed cytokines in affected subjects. These include raised levels of circulating pro-inflammatory cytokines such as IL6, IFN-γ, IL1β, IL12 as well as chemokines including CXCL10 and CCL2 (Coperchini et al., 2020). These all point to a hyperactivation of the TH1 axis of CD4 T cells. To complicate matters, it appears that SARS-CoV-2 is somewhat different to other SARS-CoV infections in that patients also demonstrate raised TH2 axis (increased IL4 and IL10 cytokines), implying that the immunosuppressive axis activates to control the overwhelming pro-inflammatory response (Zhang et al., 2020). It is an acquired response gone wild!
The most promising clinical intervention to date remains that of Zhang et al., (2020) who proposed the use of Tocilizumab (an IL6R antagonist) as a blocking agent to the binding and uptake of IL6 released by innate immune cells. This may be the breakthrough to rescuing severe patients: we await further results of this clinical study. In the interim, other authors have proposed the use of Leronimab (formerly PRO 140), a monoclonal antibody used to block the CCL5-CCR5, a pathway which seems dysregulated in Covid-19 infected patients, leading to the enhanced release of IL6 from innate immune cells (Patterson et al., 2020).
This therapy has had previous clinical application in the treatment of HIV-infected patients and was shown to inhibit the activation and chemotaxis of macrophages, the very source of the pro-inflammatory cytokines. Furthermore, Richardson et al., (2020) proposed the use of Janus kinase (JAK) inhibitors such as Baricitinib as potential therapies to control inflammation and cellular viral entry in Covid-19. Other potential drug targeting kinases have been elegantly outlined by Richardson et al., (2020) as depicted in Figure 1. In general, clinical experts suggest using approved therapies for immune suppression to prevent the cytokine storm in Covid-19 and eventual mortalities (Mehta et al., 2020).
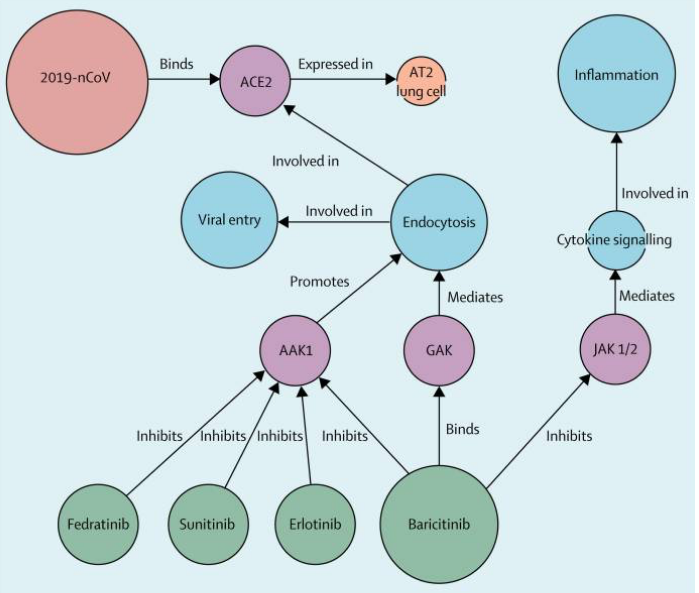
Figure 1: Taken from Richardson et al. (2020).
What then are the indicators of patients possibly needing the intensive intervention to prevent the progression from a simple “rough seas” to a full “perfect storm”? It has been a global observation that such patients all present with underlying co-morbidities such as chronic diabetes, obesity, cardio-vascular issues, and others. What makes these patients more susceptible to Covid-19 complications? The patients all present with common presentations of hypercoagulability with thrombogenesis and thromboembolism (eventually succumbing to embolism, myocardial infarction, kidney injury and strokes). He et al., (2020) recently proposed a fascinating hypothesis of the pericyte-specific vascular expression of ACE2 and its implication of microvascular inflammation. The hypothesis is illustrated below (figure 2).
The authors propose that in these pre-disposed individuals with co-morbidities, the pericytes are exposed due to low level inflammation and no longer protected by the layer of endothelial cells. These pericytes express high levels of the ACE2 receptor (the primary receptor for the virus) thereby presenting more opportunities for infection by the SARS-CoV-2, leading to increased uptake and initiation of the pathogenesis of ramped-up inflammation.
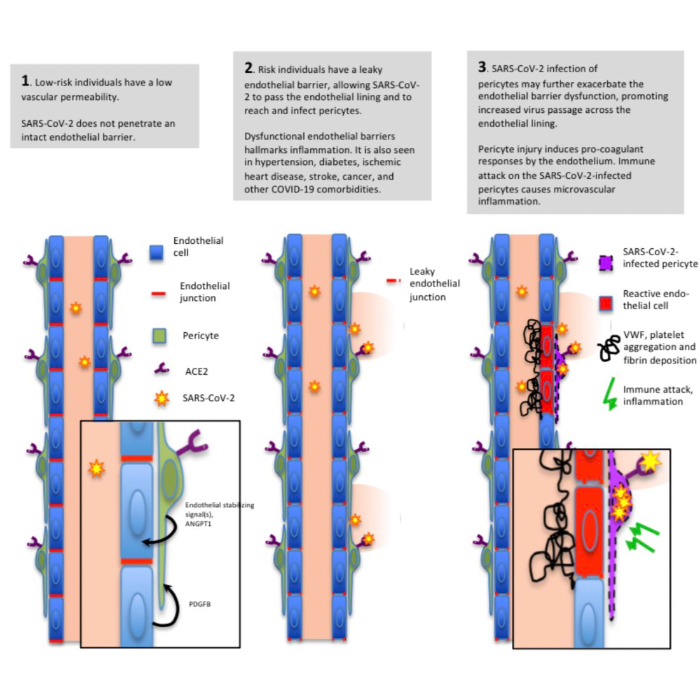
It has been suggested that due to the fact that many of these predisposed patients are treated by drugs that may ultimately lead to enhanced ACE2 receptor expression, this observation needs further investigation and clinical monitoring. Fang L, et al (2020) proposed that the use of anti-hypertensive medications, such as thiazide-type diuretics, calcium channel blockers, β-blockers and angiotensin converting enzyme inhibitors (ACEi), are the common drugs which target the control of blood pressure. Furthermore, very often these same drugs are used in the treatment of diabetes mellitus patients. However, these drugs lead to the enhanced expression of the ACE2, thereby placing the patient at an increased risk of developing more severe Covid-19 symptoms and subsequent complications: this may explain why hypertensive patients, when infected by SARS-CoV-2 are at a disadvantage and often need hospitalisation for close monitoring due to severity of symptoms.
This observation is not so conclusive since Meng et al., (2020) published their findings on the use of renin-angiotensin system inhibitors in patients with hypertension and the observed improvement in the clinical outcomes in patients with Covid-19. The interaction of such prescription drugs and clinical outcome deserves further investigation (Costa de Lucena et al., 2020).
Until the development of an effective SARS-CoV-2 drug or an effective vaccine is completed, this global pandemic is here to remain for some time to come. Subjects will become infected and hopefully the majority will recover without any significant complications. We should however as scientists be on the lookout for early biomarkers of predisposition to clinical complications and to use the current battery of drugs to prevent the cytokine storm. We need these rudders and expert ship captains to guide the ship to calmer waters.
References:
Coperchini F, Chiovato L, Croce L, Magri F & Rotondi F: https://doi.org/10.1016/j.cytogfr.2020.05.003
Zhang C, Wu Z, Li J-W, Zhoa H & Wang GQ: https://doi.org/10.1016/j.ijantimicag.2020.105954
Patterson BK, Seethamraju H, Dhody K, et al.: https://doi.org/10.1101/2020.05.02.20084673
Richardson P, Griffin I, Tucker C, et al.: https://doi.org/10.1016/S0140-6736(20)30304-4
Mehta P, McAuley DF, Brown M, et al.: https://doi.org/10.1016/S0140-6736(20)30628-0
He L, Mae MA, Sun Y, et al.: https://doi.org/10.1101/2020.05.11.088500.
Fang L, Karakiularis G & Roth M (2020): Lancet Respir Med 8, e21.
Meng J, Xiao G, Zhang J, et al.: https://doi.org/10.1080/22221751.2020.1746200.
Costa de Lucena TM, da Silva Santos AF, de Lima BR, de Albuquerque Borborema ME & de Azevedo Silva J: https://doi.org/10.1016/j.dsx.2020.05.025
